Direct Experimental Validation of Neoantigens: The Foundation of Valid-NEO®
The first prototype of what later became Valid-NEO®—a highly specialized experimental pipeline for the direct detection and quantification of neoantigens presented on HLA molecules.
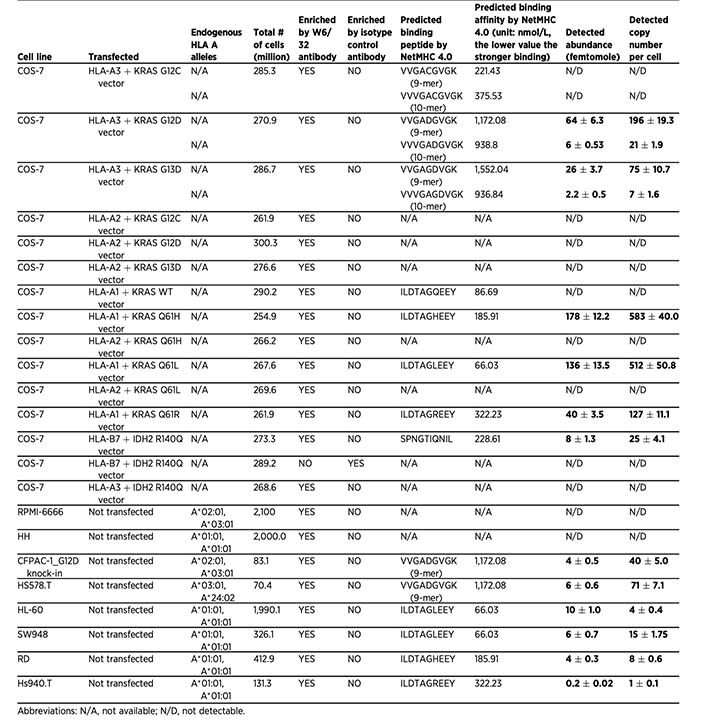
Establishing the missing experimental layer in neoantigen immunotherapy
Modern cancer immunotherapy increasingly depends on the ability to identify tumor-specific neoantigens that can be recognized by T cells. Although DNA sequencing and computational prediction made it possible to nominate large numbers of candidate neoantigens, one central problem remained unresolved: most predicted targets had never been directly confirmed to exist on the tumor cell surface.
This gap was a major translational bottleneck. A mutation may appear compelling computationally, but unless the corresponding peptide is actually processed and presented by HLA molecules, it is not a real therapeutic target. As a result, experimental validation of target presentation is one of the most important steps in rational neoantigen drug development.
This work was among the first to directly address that challenge by establishing an experimental method capable of detecting and quantifying mutation-associated neoantigens on the cell surface, rather than relying solely on prediction algorithms or indirect T-cell assays.
The prototype of Valid-NEO® analysis
This study represents the prototype of Valid-NEO® analysis and the scientific foundation of today’s Valid-NEO® assays.
The work established a highly specialized workflow that combined:
- optimized enrichment of peptide-HLA complexes
- multidimensional chromatographic separation
- recursively optimized targeted mass spectrometry
- isotope-labeled standards for direct quantification
Using this approach, the study directly confirmed neoantigens derived from major cancer driver mutations including KRAS and IDH2, and quantified the number of copies presented on the cell surface.
Importantly, the data showed that some computationally predicted neoantigens are truly presented, while others are not—despite similar in silico scores. This finding underscored a critical point that remains central today: prediction is not validation.
That principle became the core logic behind the development of Valid-NEO®.
A very high technical barrier that few groups could overcome
This work also highlighted the extraordinary technical difficulty of direct neoantigen validation.
Biologically, neoantigen-derived peptide-HLA complexes always exist at extremely low abundance, often representing only a minute fraction of all peptides presented on the cell surface. Detecting them requires overcoming multiple technical barriers simultaneously, including:
- efficient recovery of peptide-HLA complexes without major losses
- elimination of contaminating signals that suppress mass spectrometry sensitivity
- stabilization of chemically fragile peptides
- sufficient sensitivity to detect targets present at very low copy number
The study introduced a series of technical optimizations that dramatically improved performance and made direct experimental validation feasible in a way that had previously been impractical. This was not a routine proteomics workflow—it was a high-barrier immunopeptidomics method built specifically to solve one of the hardest problems in cancer immunotherapy.
That technical barrier remains one of the key reasons why validated neoantigen detection is still difficult to reproduce broadly, and why Valid-NEO® assays are strategically differentiated.
Strategic significance
This work established the core foundation for what would later become Valid-NEO®: a platform built on the principle that therapeutic targets must be experimentally verified, not just computationally predicted.
It helped define a new standard for neoantigen development by showing that direct confirmation of peptide-HLA presentation is both possible and necessary for high-confidence target selection.
As the foundation of Valid-NEO® assays, this work created the basis for a broader platform capable of supporting:
- neoantigen validation for vaccine design
- target confirmation for TCR-based therapeutics
- peptide-HLA antibody discovery
- translational immunopeptidomics from limited clinical samples
In that sense, this paper was not just an academic advance—it was the technical and conceptual starting point of the Valid-NEO® platform and a foundational step toward making neoantigen immunotherapy more precise, more rigorous, and more clinically actionable.


