Valid-NEO®: Direct Neoantigen Detection from Limited Clinical Samples
The first clinical-grade introduction of Valid-NEO®—a multi-omics platform designed to directly detect and quantify patient-specific neoantigens from very limited clinical samples.
Bringing neoantigen validation from theory into real clinical samples
Neoantigens are among the most compelling targets in modern cancer immunotherapy because they are truly tumor-specific and highly personalized. But for neoantigen-based therapies to work, one question must be answered with confidence: is the target actually presented on the patient’s tumor cell surface?
For years, this was a major limitation in the field. Most neoantigen projects depended on genomic sequencing plus algorithm-based prediction, even though prediction alone cannot determine whether a peptide is truly processed, loaded onto HLA, and presented in a clinically relevant setting. Existing mass spectrometry approaches were often impractical for real-world use, frequently requiring very large tissue inputs, invasive procedures, or workflows too fragile for routine translational application.
This paper introduced Valid-NEO® as a direct solution to that bottleneck: a platform built to move neoantigen detection from a difficult research exercise into a practical assay for clinical samples.
The introduction of the Valid-NEO® platform
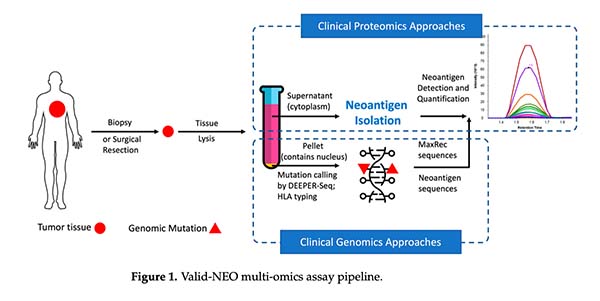
This study formally introduced Valid-NEO® as an integrated multi-omics system for the direct observation, detection, and quantification of patient-specific neoantigens from small clinical specimens.
Built on the earlier scientific foundation of our prototype neoantigen validation work, Valid-NEO advanced the field from proof-of-concept into a platform designed for real clinical use. The workflow integrated:
- rapid extraction of HLA complexes from frozen tumor tissue
- automated HLA capture and neoantigen purification
- direct targeted mass spectrometry detection
- tumor mutation profiling to define patient-specific targets
- standardized system conditioning to improve recovery and reproducibility
Most importantly, Valid-NEO was designed to detect neoantigens without relying on computational prediction as the primary evidence of target existence. Instead, the platform directly determines whether the peptide is there.
That is a major distinction. In neoantigen drug development, prediction may suggest a target, but only direct detection validates it.
A high-barrier platform built for limited samples
The technical challenge here is extreme. Neoantigens are presented at extraordinarily low abundance, often only a few copies per cell, and clinical tumor samples are usually scarce. This creates one of the hardest detection problems in translational oncology.
Valid-NEO addressed this through a series of system-level innovations, including:
- UniCeller, enabling rapid frozen-tissue disruption to single-cell-level powder while preserving HLA recovery
- an online antibody-column enrichment system for efficient HLA capture
- multi-step online purification to isolate neoantigen peptides from complex clinical matrices
- the MaxRec system, which dramatically improved recovery and sensitivity by reducing nonspecific peptide loss
- performance-standardized mass spectrometry workflows to improve assay reproducibility across runs
These innovations turned a previously fragile, labor-intensive process into a more integrated and clinically oriented assay system.
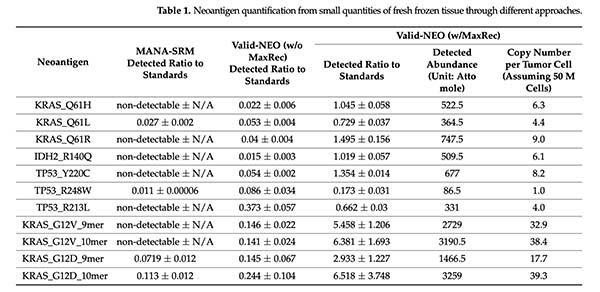
The performance gains were substantial. In this study, Valid-NEO detected neoantigens from just 5 mg of fresh frozen tumor tissue and identified targets present at very low abundance, including single-digit to low-double-digit copies per tumor cell in some cases. Compared with the earlier-generation workflow, the MaxRec-enabled Valid-NEO system delivered major improvements in sensitivity.
Strategic significance
This paper marks the formal transition of our neoantigen validation technology into the Valid-NEO® platform.
Strategically, that matters because the future of personalized immunotherapy depends not just on finding mutations, but on proving which mutated peptides are real, druggable targets. Valid-NEO was built precisely for that purpose.
The study established that direct neoantigen validation can be performed from limited clinical specimens in a way that is sensitive, rapid, and reproducible enough to support broader translational use. This creates a foundation for multiple downstream applications, including:
- personalized neoantigen vaccine design
- target validation for TCR-based therapies
- peptide-HLA antibody discovery
- companion diagnostics for neoantigen-directed therapeutics
In that sense, this paper was not simply a technical update. It was the platform introduction of Valid-NEO®—the point at which experimental neoantigen validation became positioned as a scalable, clinically relevant capability rather than a niche research method.


