March 31, 2021
Complete Omics today underscores a defining moment in precision immunotherapy: foundational neoantigen-targeting breakthroughs published in Science and Science Immunology validate one of the core scientific theses upon which the company was built.
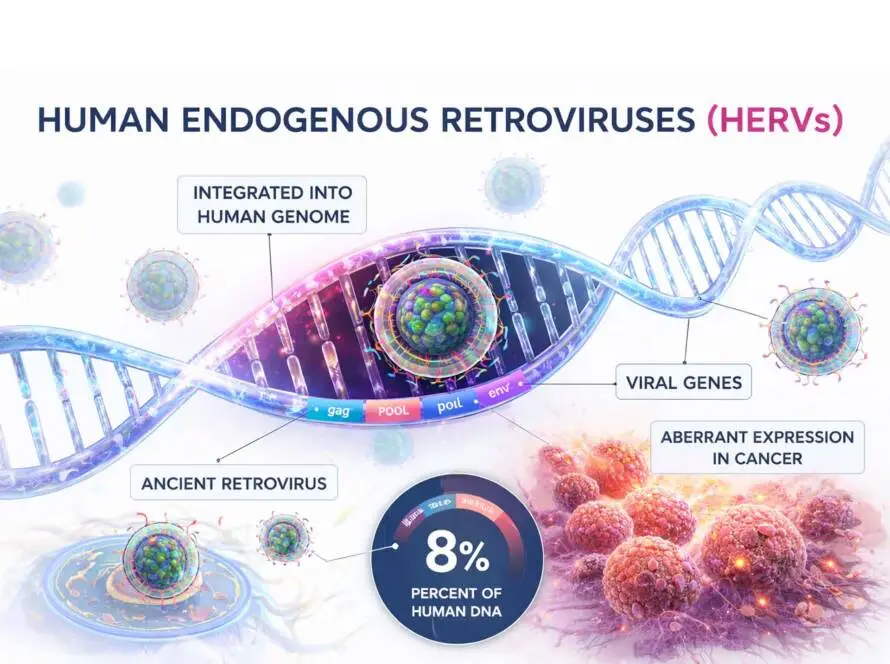
These landmark studies demonstrated that mutant KRAS and TP53 — two of the most prevalent and historically “undruggable” oncogenic drivers — can be therapeutically targeted through their HLA-presented neoantigen peptides.
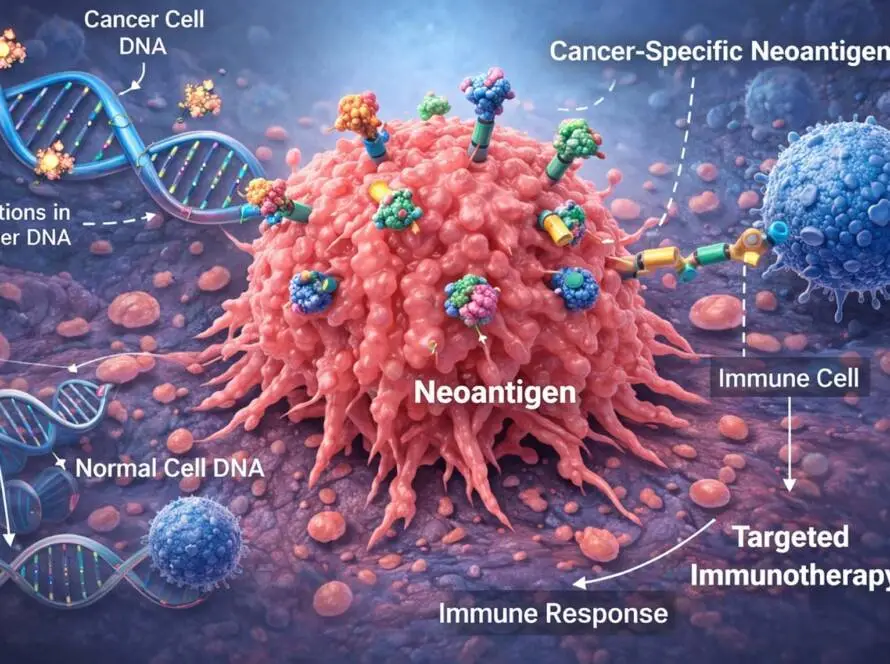
At the heart of this paradigm shift lies a fundamental requirement: ultra-sensitive, peptide-level validation of mutation-derived neoantigens.
The Paradigm Shift: From Genomic Prediction to Molecular Proof
For over a decade, oncology has operated on a predictive framework:
Mutation → Predicted Neoantigen → Therapeutic Hypothesis
But prediction is not proof. The success of neoantigen-directed antibodies, TCR therapies, and personalized cancer vaccines depends on confirming:
- The mutant protein is expressed
- The peptide is processed correctly
- It is presented by HLA molecules
- It exists at biologically actionable abundance
Without peptide-level validation, therapeutic development carries high failure risk.
The breakthroughs in Science and Science Immunology prove that common hotspot mutations such as KRAS and TP53 generate targetable neoantigens.
But they also underscore the technical barrier: detecting these peptides requires extreme sensitivity and precision.
The Complete Omics Thesis
Complete Omics neoantigen validation platform was built on a simple but powerful conviction:
The bottleneck in mutation-directed immunotherapy is not mutation discovery — it is target validation.
Founder and CEO Dr. Qing Wang pioneered ultra-sensitive mass spectrometry strategies for neoantigen detection during his faculty career at Johns Hopkins University, working in the laboratory of renowned cancer geneticist Bert Vogelstein.
His work focused on solving the exact translational gap that these Science publications now bring to the forefront: transforming genomic mutations into experimentally validated, immunologically actionable targets.

Why This Matters?
1) Intracellular Oncogenes Are Now Targetable
KRAS and TP53 were long considered beyond the reach of traditional drug modalities. Neoantigen targeting changes that equation. The addressable therapeutic universe expands dramatically when intracellular driver mutations become immunologically druggable.
2) Validation Infrastructure Becomes Mission-Critical
As bispecific antibodies, TCR therapies, and mutation-specific immunotherapies accelerate toward the clinic, demand for:
- Quantitative neoantigen confirmation
- Immunopeptidomic profiling
- Hotspot mutation abundance mapping
will grow exponentially.
Complete Omics is building the industrial-grade validation engine required to support this wave.
3) A Platform Play, Not a Single Assay
Complete Omics is not developing a single therapeutic.
It is constructing the molecular infrastructure that enables:
- Mutation-level therapeutic targeting
- Companion diagnostic strategies
- Patient stratification
- Neoantigen atlas development
This positions the company as a picks-and-shovels platform for the mutation-directed immunotherapy economy.
We are entering a category-defining era in oncology. Immunotherapy is shifting from broad immune activation to precision, mutation-directed strategies targeting shared drivers such as KRAS and TP53. These landmark publications validate a fundamental truth: mutation-derived peptides are actionable therapeutic targets. But actionability depends on measurement. At Complete Omics, we are building the ultra-sensitive measurement layer that converts genomic mutations into experimentally validated immunologic targets. This is not simply peptide detection — it is the enabling infrastructure for next-generation precision immunotherapy. Our platform is designed to identify and quantitatively measure actionable neoantigens derived from personalized cancer driver mutations with a level of sensitivity, specificity, and rigor previously unreachable in the field.
— Qing
What’s next?
Complete Omics is advancing toward:
- Large-scale neoantigen hotspot mapping
- Quantitative abundance atlases for high-frequency mutations
- Clinical-grade immunopeptidomics workflows
- Integration of genomic prediction with proteomic validation
- Strategic partnerships with immunotherapy developers
As mutation-specific therapies advance, validated peptide detection will become a core enabling technology. Complete Omics intends to define that category.
About Complete Omics—
Complete Omics is building the measurement infrastructure for the human proteome, enabling large-scale, reproducible detection of thousands of proteins from complex biological samples such as plasma. By combining advanced mass spectrometry, engineered workflows, and AI-driven analytics, the company is transforming proteomics from a research discipline into a scalable clinical platform for precision medicine. Complete Omics operates a CLIA-certified, CAP-accredited clinical laboratory, enabling the rapid translation of its technologies into deployable clinical assays. The company’s mission is to make the human proteome measurable at scale, creating a new molecular layer of medicine that powers early disease detection, therapeutic monitoring, and next-generation drug development across every disease.