October 10, 2019
Complete Omics today officially announces its founding with a bold and long-term mission:
To establish clinical proteomics as the next foundational pillar of precision medicine.
Over the past decade, genomics has transformed biomedical research and clinical oncology. Liquid biopsy technologies reshaped early cancer detection and disease monitoring. Yet genes represent biological potential. Proteins execute biological function.
Complete Omics is founded on a clear conviction:
The next revolution in precision medicine will be driven by functional, quantitative proteomics.

Originating from Johns Hopkins
Complete Omics traces its scientific roots to Johns Hopkins University School of Medicine, one of the world’s leading centers for biomedical innovation.
Founder Qing Wang, PhD, MHS began his training at Johns Hopkins as a PhD student, continued through postdoctoral research, and later served as faculty. During this time, he worked in the laboratory of Bert Vogelstein, widely recognized as the father of cancer liquid biopsy.
Dr. Vogelstein’s groundbreaking work established the scientific framework for circulating tumor DNA–based diagnostics, fundamentally changing how cancer is detected and monitored.
Working at the forefront of cancer genomics and translational oncology, Dr. Wang witnessed the power of molecular diagnostics — but also recognized a defining frontier yet to be fully unlocked:
During my years at Johns Hopkins, working in Bert’s lab, I witnessed how liquid biopsy transformed cancer diagnostics. It redefined what was possible. But I also saw the limitations of relying solely on DNA.
Proteins execute biology. Proteins determine drug response. Proteins define immune recognition.
Complete Omics was founded to build the next layer of precision medicine — one that measures disease at the functional level in a real-time, dynamic manner. Our goal is to establish clinical proteomics as a foundational technology for diagnostics and therapeutic decision-making in the coming decade.
— Qing
A Platform Company, Not Just a Service Laboratory
Complete Omics is not launching as a conventional testing lab.
We are building a platform company designed to create durable technological advantage in clinical proteomics — integrating proprietary wet-lab innovation, scalable quantitative workflows, and translational deployment capability.
Our strategy is built on two foundational pillars:
1) Liquid Biopsy: Advancing Ultra-Deep Protein Detection in Blood
Blood is the most scalable diagnostic medium in medicine — but the plasma proteome has remained one of biology’s hardest frontiers. Its extreme dynamic range and complexity have historically limited depth, sensitivity, and reproducibility. Dr. Qing Wang began working on plasma proteomics in 2007 at Johns Hopkins, and have worked through virtually every major technological paradigm for protein detection in body fluids. The bottleneck was never vision — it was performance.
Complete Omics is engineering next-generation mass spectrometry platforms to:
• Detect low- and ultra-low-abundance proteins
• Expand measurable dynamic range
• Minimize noise and matrix interference
• Deliver reproducible quantification across large cohorts
• Scale seamlessly into clinical deployment
Our ambition is not incremental — it is structural. We are building protein-based liquid biopsy as a foundational engine for early disease detection, disease monitoring, therapeutic response assessment, and precision risk stratification.

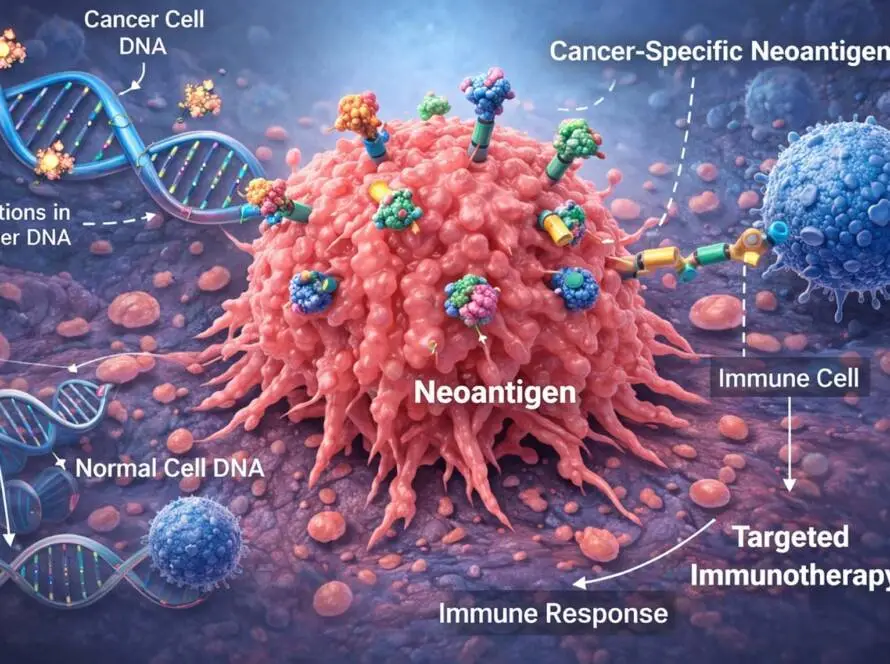
2) Tissue Biopsy: Functional Target Discovery & Personalized Therapeutics
Tumor sequencing has become standard practice. However, mutation lists alone do not reveal which targets are expressed, druggable, immunogenic, or biologically dominant.
Complete Omics is advancing tissue-based proteomics and immunopeptidomics to:
- Identify patient-specific neoantigens for personalized immunotherapy
- Detect actionable protein targets directly from tumor samples
- Characterize signaling networks and pathway activation states
- Integrate proteomic findings with genomic and transcriptomic data
Our objective is bold:
To shift personalized oncology from mutation-informed treatment selection to functionally validated therapeutic targeting.
In cancer therapy — particularly immunotherapy — direct measurement of protein expression and antigen presentation is indispensable. We aim to provide that functional clarity.
From Genomic Revolution to Functional Precision Medicine
The genomic era reshaped medicine, the proteomic era will complete it. Drawing on more than a decade of experience in large-scale cancer genomics, translational oncology, and molecular diagnostics at Johns Hopkins, Dr. Wang established Complete Omics to build the infrastructure required for the next decade of precision medicine.
By integrating:
- Ultra-deep proteomics in blood
- Neoantigen identification from tumor tissue
- Direct druggable target discovery
- Multi-omics integration across nucleic acid, protein, and metabolite.
Complete Omics seeks to define a new category: